
Currently, rapid microbial tests for pathogens are based on technologies such as immunoassays, chromatography and polymerase chain reactions. “I believe the enzyme-linked immunosorbent assays (ELISAs) and modified/adjuncts are the most commonly used methods,” says Purnendu Vasavada, Ph.D., food science professor, University of Wisconsin-River Falls. “This is due to their familiarity, convenience, regulatory and industry acceptance, and affordable cost.”
Modified/adjunct tests combine several conventional tests into convenient rapid test kits. For example, 3M's Petrifilm™ Rapid S. aureus Count Plate combines the three-plate Baird-Parker agar and tube coagulase tests for Staphylococcus aureus in a single test procedure. It consists of a sample-ready Petrifilm plate coated with modified Baird-Parker nutrients. The user places a small blue disk that has been coated with a microbial growth indicator and a thermonuclease-reactive agent within the plate's well during the final steps of the test.
Immunoassays are based on the qualitative or quantitative determination of antigen or antibody present in a sample by specific antigen-antibody reaction, explains Vasavada. Results can be visualized in terms of clumping or agglutination, color or fluorescent development from an enzyme reaction or formation of an immunoband, he says. The enzyme immunoassay (EIA) and the ELISAs are the most commonly used techniques.
Polymerase chain reaction results in the amplification of a target DNA segment. It allows for the detection of specific gene fragments by in-vitro enzymatic amplification of the target DNA, followed by detection of the amplified DNA molecule.

The Choice is Yours
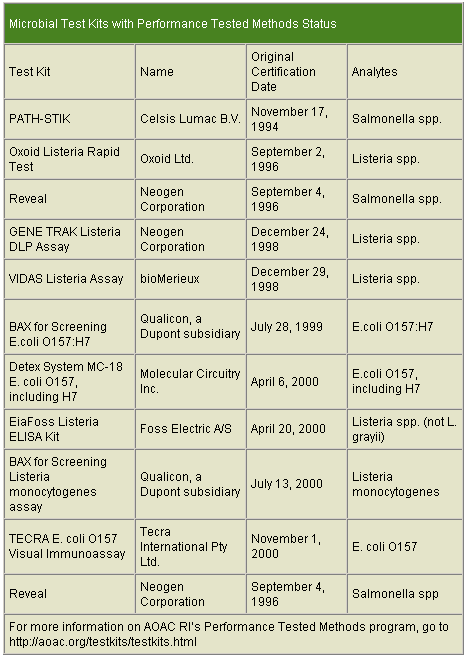
With the number of rapid tests and their various technologies, it may seem difficult to decide which test is the most appropriate.AOAC International provides one source where companies can find information on available methods that are AOAC-approved or tested by the AOAC Research Institute (AOAC RI). (See chart on p. 47) The AOAC RI is a non-profit, international, scientific organization that administers the Performance Tested Methods Program. This program provides an independent third-party review of test kit performance claims. AOAC RI grants Performance Tested Methods status to test kits found to be in conformance with their claims. The Performance Tested Methods status assures the test kit user that an independent assessment has been conducted and the kit performs as claimed.
Expert reviewers and a general referee review performance data submitted by test kit manufacturers; the test kit performance is verified at an independent testing laboratory. If all of the laboratory data supports the product performance claims, the test is granted Performance Tested Methods status. The manufacturers are then licensed to use the Performance Tested Methods certification mark on their packaging and in their advertisement. Certification must be renewed annually.
Real-Time Applications and Benefits
Obvious benefits of rapid microbial tests are time-savings, efficiency and reduced labor costs. Typically, conventional testing can take five to 10 days. Rapid tests, however, can take anywhere from a day to a day-and-a-half to detect the presence of pathogens.The immunoassays and adjunct test kits are the most widely used in the food industry, perhaps due to cost factors. PCR techniques, however, are making inroads. DuPont's Qualicon business, Wilmington, Del., has established a collaboration with the Antimicrobial Resistance Research Unit of the U.S Department of Agriculture (USDA) to develop and evaluate a PCR-based assay for the detection and speciation of Campylobacter jejuni and E. coli. The test will run on Qualicon's BAX system platform for pathogen detection. The system makes PCR extremely easy to use by combining all necessary reagents and a positive control into a prepackaged tablet. In addition to the development of the assay, the work will include a cooperative project with the USDA Agricultural Research Service (ARS) and Food Safety and Inspection Service (FSIS) to make a population study of Campylobacter contamination of poultry carcasses and evaluation of the BAX Campylobacter assay for screening isolates processed through the National Antimicrobial Resistance Monitoring Survey (NARMS).
FSIS is currently evaluating the BAX system to screen ready-to-eat products for Listeria monocytogenes. If implemented, it could reduce the reporting time for negative L. monocytogenes samples by one day and negative Salmonella samples by two days. When used to screen for E. coli O157:H7, the BAX system may take several hours longer, but it could greatly reduce the number of false positives.
Where Do We Go From Here?
Although testing for pathogens has evolved, there is still room for improvement. “Two areas are important: one is requirement for enrichment to detect a few pathogens; second is resuscitation of injured organisms and viable but non-culturable cells,” says Vasavada.The Department of Food Science & Technology, Mississippi State University, addressed the question of being able to differentiate viable cells from nonviable cells in molecular-based detection techniques such as PCR. At this year's IFT Food Expo, students presented information on a technique called quantitative competitive PCR (QC-PCR) that employs a competitive DNA sequence as an internal standard in PCR reactions. This technique could be used at multiple sampling points in an enrichment to detect and confirm the presence of viable target cells. Their work showed that QC-PCR could be applied to detect and confirm the presence of viable E. coli O157:H7 in broth or ground meat.
The Department of Nutrition & Food Sciences, Utah State University, developed a detection method utilizing specific antibodies for rapid capture of pathogens that does not require pre-enrichment. In their research, they attached monoclonal antibodies against E. coli O157:H7 to chemically modified glass beads. They inoculated apple juice and beer with E. coli O157:H7 at 105 cfu/25 mL and 50 mL aliquots of the food samples were incubated with the modified antibody beads. Capture and identification of bound organisms was detected spectrophotometrically after the addition of enzyme labeled antibodies and substrate. The total length of the test, from sample preparation to results, was 2.5 hours.
The detection limit was 100 cfu/25 mL of apple juice and beer. The work indicates that it would be possible to shorten the length of time involved for pathogen testing when the pre-enrichment step is omitted using their test.
"Current Concepts In Foodborne Pathogens and Rapid and Automated Methods in Food Microbiology" will be held October 13-16, 2002 at the University of Wisconsin-River Falls. For more information, go to: www.uwrf.edu/food-science/foodmicr.htm